The ADSC hosted an exciting webinar showcasing two projects funded by the Innovation to Improve Ability program at the Deshpande Center for Technical Innovation at MIT.
Watch the full Webinar below!

The ADSC hosted an exciting webinar showcasing two projects funded by the Innovation to Improve Ability program at the Deshpande Center for Technical Innovation at MIT.
Watch the full Webinar below!

The ADSC presents an update on our Technology to Improve Ability Grant program. These talks will be accessible to the general public.
(Ravi Rasalingam and Tarsha Ward of the Ellen Roche group)
&
(Kristina Johnson and Jaya Narain of the Pattie Maes and Rosalind Picard groups)

Tuesday, July 28, 2020 10-11am EST
As people with Down syndrome age, the risk of developing Alzheimer’s disease (AD) increases significantly. The Tsai Lab discovered that by boosting gamma power, we could reverse AD related pathology and improve memory in mouse models of AD & DS. We developed a device that boosts gamma power in people using light and sound to improve memory in AD patients and individuals with Ds. In addition, the lab is using human pluripotent stem cells to generate the different types of cells in the brain, to study the mechanisms of how Trisomy 21 has such a large impact on brain function.
This webinar will not be recorded, so please plan to join live.
Diane Chan, MD PhD, Neurologist, Massachusetts Institute of Technology/Massachusetts General Hospital; Brennan Jackson, BS, Doctoral Student, Massachusetts Institute of Technology; Hiruy Meharena, PhD, Alana Senior Fellow, Massachusetts Institute of Technology
https://attendee.gotowebinar.com/register/5669979562576345359

By developing a lab-engineered model of the human blood-brain barrier (BBB), neuroscientists at MIT’s Alana Down Syndrome Center have discovered how the most common Alzheimer’s disease risk gene causes amyloid protein plaques to disrupt the brain’s vasculature and showed they could prevent the damage with medications already approved for human use.
About 25 percent of people have the APOE4 variant of the APOE gene, which puts them at substantially greater risk for Alzheimer’s disease. Almost everyone with Alzheimer’s, and even some elderly people without, suffer from cerebral amyloid angiopathy (CAA), a condition in which amyloid protein deposits on blood vessel walls impairs the ability of the BBB to properly transport nutrients, clear out waste and prevent the invasion of pathogens and unwanted substances.
In the new study, published June 8 in Nature Medicine, the researchers pinpointed the specific vascular cell type (pericytes) and molecular pathway (calcineurin/NFAT) through which the APOE4 variant promotes CAA pathology.
The research indicates that in people with the APOE4 variant, pericytes in their vessels churn out too much APOE protein, explained senior author Li-Huei Tsai, Picower Directory & Professor of Neuroscience and co-Director of the Alana Down Syndrome Center. APOE causes amyloid proteins, which are more abundant in Alzheimer’s disease, to clump together. Meanwhile, the diseased pericytes’ increased activation of the calcineurin/NFAT molecular pathway appears to encourage the elevated APOE expression.
There are already drugs that suppress the pathway. Currently they are used to subdue the immune system after a transplant. When the researchers administered some of those drugs, including cyclosporine A and FK506, to the lab-grown BBBs with the APOE4 variant, they accumulated much less amyloid than untreated ones did.
“We identify that there is a specific genetic pathway that is expressed differently in a population that is susceptible to Alzheimer’s disease,” said study lead author Joel Blanchard, a postdoc in Tsai’s lab. “By identifying this we could identify drugs that change this pathway back to a non-diseased state and correct this outcome that’s associated with Alzheimer’s.”
To investigate the connection between Alzheimer’s, the APOE4 variant and CAA, Blanchard, Tsai and co-authors coaxed human induced pluripotent stem cells to become the three types of cells that make up the BBB: brain endothelial cells, astrocytes and pericytes. Pericytes were modeled by mural cells that they tested extensively to ensure they exhibited pericyte-like properties and gene expression.
Grown for two weeks within a three-dimensional hydrogel scaffold, the BBB model cells assembled into vessels that exhibited natural BBB properties, including low permeability to molecules and expression of the same key genes, proteins and molecular pumps as natural BBBs. When immersed in culture media high in amyloid proteins, mimicking conditions in Alzheimer’s disease brains, the lab-grown BBB models exhibited the same kind of amyloid accumulation seen in human disease.
With a model BBB established, they then sought to test the difference APOE4 makes. They showed by several measures that APOE4-carrying BBB models accumulated more amyloid from culture media than those carrying APOE3, the more typical and healthy variant.
To pinpoint how APOE4 makes that difference, they engineered eight different versions covering all the possible combinations of the three cell types having either APOE3 or APOE4. When exposed these month-old models to amyloid-rich media, only versions with APOE4 pericyte-like mural cells showed excessive accumulation of amyloid proteins. Replacing APOE4 mural cells with APOE3-carrying ones reduced amyloid deposition. These results put blame for CAA-like pathology squarely on pericytes.
To further validate the clinical relevance of these findings, the team also looked at APOE expression in samples of human brain vasculature in the prefrontal cortex and the hippocampus, two regions crucially affected in Alzheimer’s disease. Consistent with the team’s lab BBB model, people with APOE4 showed higher expression of the gene in the vasculature, and specifically in pericytes, than people with APOE3.
“That is a salient point of this paper,” said Tsai. “It’s really cool because it stresses the cell-type specific function of APOE.”
The next step was to determine how APOE4 becomes so overexpressed by pericytes. The team therefore identified hundreds of transcription factors – proteins that determine how genes are expressed – that were regulated differently between APOE3 and APOE4 pericyte-like mural cells. Then they scoured that list to see which factors specifically impact APOE expression. A set of factors that were upregulated in APOE4 cells stood out: ones that were part of the calcineurin/NFAT pathway. They observed similar upregulation of the pathway in pericytes from human hippocampus samples.
As part of their investigation of whether elevated signaling activity of this pathway caused increased amyloid deposition and CAA, they tested cyclosporine A and FK506 because they tamp pathway activity down. They found that the drugs reduced APOE expression in their pericyte-like mural cells and therefore APOE4-mediated amyloid deposits in the BBB models. They also tested the drugs in APOE4-carrying mice and saw that the medicines reduced APOE expression and amyloid buildup.
Blanchard and Tsai noted that the drugs can have significant side effects, so their findings might not suggest using exactly those drugs to address CAA in patients.
“Instead it points toward the value of understanding the mechanism,” Blanchard said. “It allows one to design a small molecule screen to find more potent drugs that have less off-target effects.”
In addition to Blanchard and Tsai, the paper’s other authors are Michael Bula, Jose Davila-Velderrain, Leyla Akay, Lena Zhu, Alexander Frank, Matheus Victor, Julia Maeve Bonner, Hansruedi Mathys, Yuan-Ta Lin, Tak Ko, David Bennett, Hugh Cam, and Manolis Kellis.
The Robert A. and Renee E. Belfer Family Foundation, the Cure Alzheimer’s Fund, The National Institutes of Health, the Glenn Foundation for Medical Research and the American Federation for Aging Research funded the research.
Written by David Orenstein, Picower Institute for Learning and Memory
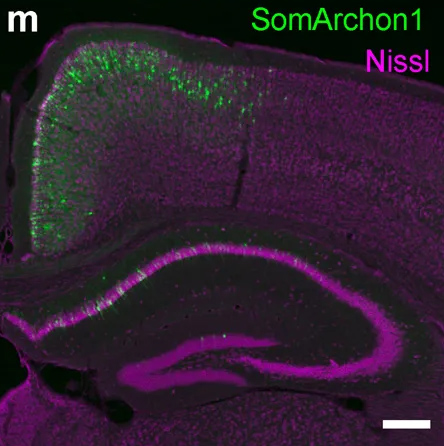
Using a fluorescent probe that lights up when brain cells are electrically active, MIT and Boston University researchers have shown that they can image the activity of many neurons at once, in the brains of mice.
This technique, which can be performed using a simple light microscope, could allow neuroscientists to visualize the activity of circuits within the brain and link them to specific behaviors, says Edward Boyden, the Y. Eva Tan Professor in Neurotechnology and a professor of biological engineering and of brain and cognitive sciences at MIT.
“If you want to study a behavior, or a disease, you need to image the activity of populations of neurons because they work together in a network,” says Boyden, who is also a member of MIT’s McGovern Institute for Brain Research, Media Lab, and Koch Institute for Integrative Cancer Research, and is a member of the Alana Down Syndrome Center.
Using this voltage-sensing molecule, the researchers showed that they could record electrical activity from many more neurons than has been possible with any existing, fully genetically encoded, fluorescent voltage probe.
Boyden and Xue Han, an associate professor of biomedical engineering at Boston University, are the senior authors of the study, which appears in the Oct. 9 online edition of Nature. The lead authors of the paper are MIT postdoc Kiryl Piatkevich, BU graduate student Seth Bensussen, and BU research scientist Hua-an Tseng.
Neurons compute using rapid electrical impulses, which underlie our thoughts, behavior, and perception of the world. Traditional methods for measuring this electrical activity require inserting an electrode into the brain, a process that is labor-intensive and usually allows researchers to record from only one neuron at a time. Multielectrode arrays allow the monitoring of electrical activity from many neurons at once, but they don’t sample densely enough to get all the neurons within a given volume. Calcium imaging does allow such dense sampling, but it measures calcium, an indirect and slow measure of neural electrical activity.
In 2018, Boyden’s team developed an alternative way to monitor electrical activity by labeling neurons with a fluorescent probe. Using a technique known as directed protein evolution, his group engineered a molecule called Archon1 that can be genetically inserted into neurons, where it becomes embedded in the cell membrane. When a neuron’s electrical activity increases, the molecule becomes brighter, and this fluorescence can be seen with a standard light microscope.
In the 2018 paper, Boyden and his colleagues showed that they could use the molecule to image electrical activity in the brains of transparent worms and zebrafish embryos, and also in mouse brain slices. In the new study, they wanted to try to use it in living, awake mice as they engaged in a specific behavior.
To do that, the researchers had to modify the probe so that it would go to a subregion of the neuron membrane. They found that when the molecule inserts itself throughout the entire cell membrane, the resulting images are blurry because the axons and dendrites that extend from neurons also fluoresce. To overcome that, the researchers attached a small peptide that guides the probe specifically to membranes of the cell bodies of neurons. They called this modified protein SomArchon.
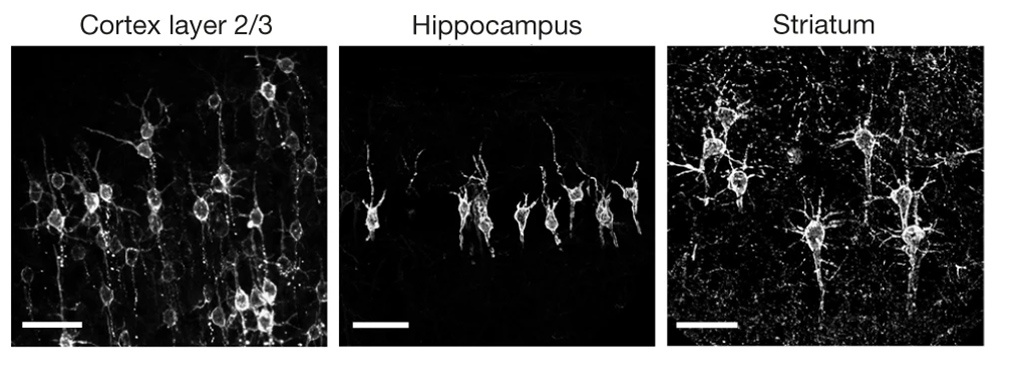
“With SomArchon, you can see each cell as a distinct sphere,” Boyden says. “Rather than having one cell’s light blurring all its neighbors, each cell can speak by itself loudly and clearly, uncontaminated by its neighbors.”
The researchers used this probe to image activity in a part of the brain called the striatum, which is involved in planning movement, as mice ran on a ball. They were able to monitor activity in several neurons simultaneously and correlate each one’s activity with the mice’s movement. Some neurons’ activity went up when the mice were running, some went down, and others showed no significant change.
“Over the years, my lab has tried many different versions of voltage sensors, and none of them have worked in living mammalian brains until this one,” Han says.
Using this fluorescent probe, the researchers were able to obtain measurements similar to those recorded by an electrical probe, which can pick up activity on a very rapid timescale. This makes the measurements more informative than existing techniques such as imaging calcium, which neuroscientists often use as a proxy for electrical activity.
“We want to record electrical activity on a millisecond timescale,” Han says. “The timescale and activity patterns that we get from calcium imaging are very different. We really don’t know exactly how these calcium changes are related to electrical dynamics.”
With the new voltage sensor, it is also possible to measure very small fluctuations in activity that occur even when a neuron is not firing a spike. This could help neuroscientists study how small fluctuations impact a neuron’s overall behavior, which has previously been very difficult in living brains, Han says.
The researchers also showed that this imaging technique can be combined with optogenetics— a technique developed by the Boyden lab and collaborators that allows researchers to turn neurons on and off with light by engineering them to express light-sensitive proteins. In this case, the researchers activated certain neurons with light and then measured the resulting electrical activity in these neurons.
This imaging technology could also be combined with expansion microscopy, a technique that Boyden’s lab developed to expand brain tissue before imaging it, make it easier to see the anatomical connections between neurons in high resolution.
“One of my dream experiments is to image all the activity in a brain, and then use expansion microscopy to find the wiring between those neurons,” Boyden says. “Then can we predict how neural computations emerge from the wiring.”
Such wiring diagrams could allow researchers to pinpoint circuit abnormalities that underlie brain disorders, and may also help researchers to design artificial intelligence that more closely mimics the human brain, Boyden says.
The MIT portion of the research was funded by Edward and Kay Poitras, the National Institutes of Health, including a Director’s Pioneer Award, Charles Hieken, John Doerr, the National Science Foundation, the HHMI-Simons Faculty Scholars Program, the Human Frontier Science Program, and the U.S. Army Research Office.
By
Ann Trafton, MIT News Office

At its first ever symposium Nov. 6, the Alana Down Syndrome Center demonstrated ways, from the scale of chromosomes to that of caregiving communities, that scientists and physicians in Massachusetts and around the country are working to help people with Down syndrome live their healthiest, fullest lives.
Founded at MIT in March 2019, the ADSC brings together neuroscience, biology, engineering and computer science research labs together with the Desphande Center for Technological Innovation to deepen knowledge about Down syndrome and to improve health, autonomy and inclusion of people with the genetic condition characterized by an extra copy of chromosome 21.
The symposium, “Translational Research in Down Syndrome,” brought together experts working across a spectrum of fundamental biology to clinical care. In her opening remarks, ADSC co-director Li-Huei Tsai, Picower Professor of Neuroscience and director of The Picower Institute for Learning and Memory, said the event represented a chance for conversation and collaboration among researchers with the common goal of helping people with Down syndrome.
“Informed and inspired by their remarks we can all engage today in learning from each other,” she said. Tsai also thanked Ana Lucia Villela, whose Alana Foundation gift established the center and who had returned to MIT from Brazil to attend the symposium.
Throughout the afternoon, speakers shared some of their latest insights into how “aneuploidy,” having an atypical number of chromosomes, alters the biology of cells, the body and the brain.
One consequence appears to be that with an extra chromosome, cells make too many copies of the protein subunits that the chromosome encodes. Normally these subunits would become bound with partners encoded elsewhere into larger protein complexes, said ADSC Co-Director Angelika Amon, Kathleen and Curtis Marble Professor of Cancer Research in MIT’s biology department and the Koch Center for Integrative Cancer Research. But there aren’t as many of those partners, so the excess, unbound proteins become prone to clumping together, creating a major clean-up job for the cells that causes ”proteotoxic” stress. In Down syndrome, she said, that stress can hinder growth and proper function. Aneuploidy, she added, might also lead to a greater incidence of DNA damage.

Former Amon lab postdoc Eduardo Torres, who is now at the University of Massachusetts Medical School, said his lab has found that aneuploidy also disrupts the very shape and structure of the nucleus in a variety of cells, making them more sensitive to mechanical stress. The lab looked deeper to find the genetic and molecular pathway responsible and identified one related to the lipid composition of the nucleus. That insight allowed them to discover that administering certain drugs to cells with aneuploidy of chromosome 21 (or 13 or 18) can help shore up the nuclear structure and help cells grow.
To gain more insight into how aneuploidy affects neurological development many scientists have begun using techniques to grow brain cells from stem cells derived from Down syndrome patients. They can manipulate these cultures in the lab so that the only genetic difference is the extra copy of chromosome 21. Jeanne Lawrence, also of UMass, said use of such advanced models will help her understand whether a technique her lab has developed to silence extra copies of a chromosome will be effective in cells such as those in the brain or blood. Her work shows promise for a potential gene therapy to mitigate the effects of the extra copy of chromosome 21.
Another vital model of Down syndrome is the mouse. In one of the day’s two keynote addresses, Roger Reeves of the McKusick-Nathans Institute for Genetic Medicine at Johns Hopkins University described what researchers have learned from the widely used T65dn mouse model, as well as what they hope to learn from a newly developed model, that uses human chromosome 21 genes to replicate chromosome duplication. He also described their studies of the developmental anomalies in Down syndrome model mouse brains, and have found that a crucial signaling pathway for development is less responsive in these mice. He reported the results of a screen to look for the specific contributors this pathway in DS, as they may be viable targets for drug development, and his lab has also identified some of these same genes to be involved in congenital heart defects.
In the symposium’s other keynote, Joaquin Espinosa of the Linda Crnic Institute for Down Syndrome at the University of Colorado, discussed how a fast-emerging raft of insights including discoveries about the immune system in Down syndrome has led to a new clinical trial. Fundamental research at the institute has found that patients with Down syndrome have an increased sensitivity to interferons, proteins emitted by immune cells as they fight infections. The research led scientists to test medicines to calm the immune system. He described their current work on a clinical trial that aims to investigate a drug, Xeljanz, already used for auto-immune disease, to see if the drug not only improves autoimmune skin diseases, but possibly a wider range of symptoms associated with Down syndrome.
Another clinical trial is getting underway at Boston Children’s Hospital, said Nicole Baumer, a researcher there who said there are real opportunities for interventions to improve cognition in Down syndrome patients, but who also cautioned that researchers must always consult patients and their caregivers about what they want from clinical trials and care, rather than assuming what’s best for them. After surveying to learn more about patient and family wishes, her group has designed a study in which they will try to predict the neurodevelopmental outcomes in babies with Down syndrome, and test whether behavioral therapy interventions designed for certain autism populations might also augment intellectual development in children with Down syndrome.

As researchers strive in the lab and clinic to make new discoveries and improve care, Brian Skotko of Massachusetts General Hospital and Harvard Medical School has also been considering how to ensure that state-of-the-art information reaches doctors and family caregivers everywhere it’s needed. Skotko noted that among approximately 212,000 people with Down syndrome in the United States, less than five percent have access to one of the 71 specialty clinics around the country like the one he directs at MGH. Instead, they typically depend on primary care physicians. That’s why he and a diverse team have spent the last two years developing an Internet-based platform, “Down Syndrome Clinic to You (DSC2U)” in which a physician or other caregiver can enter information about a patient and learn richly linked, expert-curated information and recommendations about medical care and wellness customized for the entered patient profile. The clinical team at MGH reviews the underlying database regularly to keep it up to date. With new data showing that the system positively influences care and has been valued by users, it’s ready for a wider launch next year, he said.
Taken together, the symposium talks illustrated many routes to potential progress, from the cell to the clinic.
Session 443: Oct 22, 12 – 1:10pm, Hall B
| Hiruy Meharena | Tsai lab |
Altered 3D-Genome Architecture of Neural Progenitor Cells as a Consequence of Down Syndrome |
Session 113.26 8 AM- 12 PM |
| Y.-T. Lin | Tsai lab |
Engineered human cerebral organoids as a model for studying Down Syndrome |
Session 278.19 8 AM- 12 PM |
| C. Addaikkan | Tsai lab |
Identifying neural circuits underlying visually evoked entrainment |
Session 577.08 1 PM- 5 PM |
| A. Payne | Boyden lab |
Genome-wide in situ sequencing |
Session 611.05 1 PM- 5 PM |
| A. Sinha | Boyden lab |
Targeted and untargeted in situ sequencing in thick, physically magnified brain tissue |
Session 611.06 1 PM- 5 PM |
| E.R. Lockshin | Tsai lab |
Glial cell dysfunction as a result of 3D genome architecture changes in risomy-21 iPSC and ts65DN mouse models |
Session 729.08 1 PM- 5 PM |
| B. Jackson | Tsai & Boyden labs |
Extended gamma sensory stimulation in cognitively normal individuals |
Session 771.06 1 PM- 5 PM |
| D. Sarkar | Boyden lab |
Iterative direct expansion microscopy |
Session 794.08 1 PM- 5 PM |

The ADSC at MIT welcomes you to our first annual Down Syndrome symposium, Translational Research in Down Syndrome.
Wednesday, November 6
1:00 – 6:00pm
MIT Building 46
43 Vassar St, 46-3002
3rd Floor, Singleton Auditorium
Keynote speakers:
Roger Reeves, McKusick-Nathans Institute for Genetic Medicine, Johns Hopkins University
Joaquin Espinosa, Linda Crnic Institute for Down Syndrome, University of Colorado
Additional Speakers:
Angelika Amon, MIT, Alana Down Syndrome Center
Nicole Baumer, Boston Children’s Hospital
Jeanne Lawrence, University of Massachusetts Medical School
Brian Skotko, Massachusetts General Hospital
Eduardo Torres, University of Massachusetts Medical School